Challenges

The “Health and Wellbeing” theme is part of the ten major challenges to society currently identified by the French Ministry of Higher Education and Research’s new SNR national research strategy, closely linked to Europe’s Horizon 2020 innovation and research programme. Health is a strategic development factor both as regards its related economic activity and the expectations of each citizen. Society expects health risks to be kept under control as this is a condition for personal wellbeing and development. The economic stakes involved in animal and plant health are high. The challenge for research is thus to help prevent, detect and treat diseases affecting human, animal or plant health.

These include infectious diseases caused by a pathogenic microorganism such as a virus, bacterium, parasite or fungus, one of the key causes of death worldwide and responsible for nearly one third of deaths in low-income countries (World Health Organisation). The pathogens responsible for these diseases may be acquired by direct contact with an infected reservoir such as water (e.g. cholera or bilharzia), with a host such as humans (e.g. meningitis), or by the bite or sting of a vector insect (e.g. malaria, dengue fever, chikungunya, leishmaniasis, Rift Valley fever, Lyme’s disease and West Nile fever). The combat against such diseases—involving epidemiological surveillance and the development of treatment and vaccines—therefore remains topical in a context of rapid environmental change (climate change, population growth, deforestation and urbanisation, intensification of farming practices, globalisation and the intensification of trading etc.) which fosters the maintenance and spread of pathogens, thus contributing to the endemic or emerging nature of diseases in humankind, wildlife, domestic animals and plants. Non-communicable diseases such as respiratory disease, cardiovascular disease or cancer are also responsible for a growing number of deaths worldwide (World Health Organisation), possibly caused by organic or inorganic elements in the environment (e.g. atmospheric pollution, accentuated by climate change and the densification of urban populations; or the content of methylmercury in rivers, naturally present in certain areas of the world, but increased by deforestation and certain gold-mining processes).
In the light of these challenges, an integrated approach is being developed to investigate the close yet complex relations between the environment, ecosystems and aetiological agents responsible for disease in human, animal and plant populations focusing on ‘One Health’ and ‘EcoHealth’. This research needs to clarify the mechanisms at work and identify factors that foster the appearance, spread and persistence of disease. These factors may be environmental, climatic, demographic, socio-economic or behavioural. Some may be identified from space, but this requires developing efficient methods to turn remote sensing into an appropriate tool for characterizing, mapping and monitoring these risk factors: this is the challenge of the “Remote Sensing and Health” theme.
Research issues
Research issues related to the “remote sensing and health” theme concern the whole information processing chain, from data acquisition to prevention and disease control. They may be divided into three main groups:
1) How can we characterise situations and phenomena of relevance to the study of eco-pathosystems and health management? From the initial applications of remote sensing to health in the 1970s, the scientific community has acquired a certain experience on the characterisation of environmental conditions that foster the transmission or spread of diseases with environmental determinants by directly identifying, for example, the presence of pathogenic agents (e.g. atmospheric particles) or characterising the conditions favourable to vectors, wild reservoirs, sensitive host populations (whether human, animal or plant) and their interactions. However, research efforts remain necessary to ensure greater objectivity in the choice of parameters to be characterised and the way they are characterised, particularly as technological progress is opening up access to greater spatial and temporal resolutions, requiring a constant reassessment of these research issues. The specific questions to be answered are:
- Which are the most suitable sensors and methods for processing imagery, changing scales or integrating multi-source data? Which are the most relevant satellite environmental products available (temperature, humidity, aerosols, surfaces, etc.)?
- How can we use remote sensing to characterise and monitor the spatio-temporal evolution of determinants that can foster the appearance and spread of disease?
- How can we use remote sensing to characterise the spatial distribution of socio-demographic and behavioural situations so as to identify and characterise the most vulnerable populations? While very-high-resolution sensors appear useful in this context, by enabling researchers to precisely characterise the human occupation modes of the regions being studied, little work has been carried out to address this particular issue.
2) How can we simultaneously analyse complex data, i.e. multi-source, multi-resolution, multi-scale, quantitative, qualitative, inaccurate or uncertain data with different time and space references, in order to:
- describe and explain transmission and dissemination processes?
- construct models so as to:
- construct relevant environmental indicators with which to study eco-pathosystems and health management?
- carry out simulations aimed, for instance, at testing control strategies or change scenarios and/or forecasting high-risk zones or periods for the transmission or dissemination of pathogens?
3) How can we improve the integration of expert and multi-disciplinary knowledge in order to:
- better target the situations and phenomena of interest to study health issues?
- guide the characterisation of these situations and phenomena of interest, and construct models that are plausible from a biological, ecological, socio-demographic and behavioural viewpoint?
- guide end users in the analysis and interpretation of data, indicators and model outputs?
All these questions are directly linked to the major challenge to research in this area: facilitating the appropriation and exploitation of remote sensing data, risk indicators, model outputs etc. by end users (whether researchers, health managers, the general public or others) and participating more directly in the implementation of prevention and control strategies.
Related Theia Products
Concerned Theia SECs
Theia News on Health
Applications
ANISETTE | ANalyse Inter-Site : Evaluation de la Télédétection comme ouTil prédictif pour la surveillance et le contrôle de maladies causées par des moustiquEs. (2019-2021)

https://anisette.cirad.fr
The main objective of the ANISETTE project is to assess the interoperability of the different approaches developed by the partner teams for the prediction of zones favorable to mosquito development, through inter-site analysis: for different geographical sites and mosquito species.
Specific objectives are the following:
• Identifying the Earth Observation images best suited to the development of predictive tools for mosquito population dynamics and vector-borne disease transmission, in terms of spatial, temporal and spectral resolutions, as well as their complementarities. The analysis will in particular focus on the potential of time series of images (Sentinel-2, MODIS) and their complementarity with other types of sensors (radar – Sentinel-1, thermal- Landsat8) and optical images at higher spatial resolution (SPOT 6/7, Pleiades).
• Analyzing the potential as well as the limits of existing modelling approaches (data-based, knowledge-based, process-based models) ;
• Dissemination and transfer of validated methods.
Our study model will be based on Aedes mosquitoes (dengue and Rift-Valley fever vector) and Anopheles mosquitoes (malaria vector).
Partners: Cirad | Tetis |Astre | IRD | espaceDEV | CNRS | IDEES | Institut Pasteur | CNES
Financing: CNES
Publications: In a Remote sensing special issue « Remote Sensing for Health: from Fine-Scale Investigations towards Early-Warning Systems » (Editor: V. Herbreteau)
Tran A, Fall AG, Biteye B, Ciss M, Gimonneau G, Castets M, Talla Seck M, Chevalier V. 2019. Spatial modeling of mosquito vectors for rift valley fever virus in northern Senegal: Integrating satellite-derived meteorological estimates in population dynamics models. Remote Sensing, 11 (9) (1024) : 24 p.
Sentinel-2 for Malaria surveillance (S2-Malaria) (08/2017-07/2020)
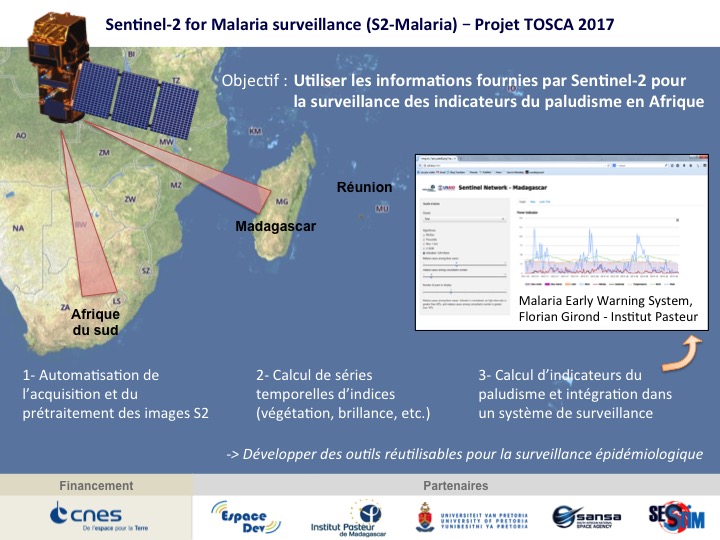
The main objective of the S2-Malaria project is to use the information provided by the Sentinel-2 satellites for malaria surveillance in Africa. Data from the acquisition and processing of Sentinel-2 images by the project will be: 1) used as a source of environmental information in a malaria surveillance system and 2) made available as time series of simple spectral indices at study sites for end users (initially in Madagascar and South Africa). The project has three methodological sub-objectives:
- adapt existing chains for the acquisition and pre-processing of Sentinel-2 images to the project. The sen2chain chain was thus produced to facilitate image downloading and processing from L1C to L2A level, index production and time series calculation (https://framagit.org/jebins/sen2chain);
- Integrate the acquisition chain and then the calculation of the indices into a monitoring system;
- Test the spectral indices already in use (from other sensors) and create other indices relevant for malaria surveillance (using the spectral richness of Sentinel-2).
In the longer term, the objective is to make these image products easily usable for epidemiological surveillance but also to multiply the uses where time series with high spatial resolution are necessary.
Location: Madagascar, South Africa
Partners: UMR Espace-Dev – IRD, Institut Pasteur de Madagascar, UMR SESSTIM – IRD, University of Pretoria, CNES
Funding: CNES TOSCA
Reference: Herbreteau Vincent, Révillion Christophe, Wolff Charlotte, Monitoring malaria in Madagascar through satellites,[IGN Mag] 88 (2017).
Website : Chain of chains for processing Sentinel-2 image series https://framagit.org/jebins/sen2chain
ECOMORE 2 | ECOnomic development, ECOsystem MOdifications, and emerging infectious diseases Risk Evaluation (2018-2020)
The ECOMORE 2 project aims to study the impact of ecosystem changes on the health of populations as a result of human activity (urbanization, intensification of agricultural techniques, land use, population movements) and climate change. It is financed by AFD and is in line with the strong international and regional concerns raised by COP21. A climate-environment component of the ECOMORE2 project proposes to set up a climate service to map the risk of transmission of dengue fever and leptospirosis in partner countries, which have experienced several epidemics over the past decade. The objective is to estimate the evolution of these risks in the context of climate change, using the IPCC (Intergovernmental Panel on Climate Change) multi-model climate data for different emission scenarios to determine whether dengue fever, leptospirosis and vectors can spread beyond their current borders. Locally, the “climate-environment” component will also aim to analyse spatially the epidemiological and entomological surveillance surveys carried out in each country.
Location: Cambodia, Laos, Myanmar, Vietnam
Partners: UMR Espace-Dev – IRD, UMR LOCEAN – IRD and UMR TETIS – CIRAD, for the “climate-environment” component of the Ecomore 2 project.
Financing : Agence Française de Développement (AFD)
Website : http://www.ecomore.org/
TEMPO project: Remote sensing and spatial modelling of animal mobility | Application to the study of domestic fauna/wildlife contacts and the risk of pathogen transmission (2018-2021)

Three components structure this project: (1) the characterization and monitoring by remote sensing of the environmental determinants of land use and movements of wild and domestic animals; (2) the modelling of land use dynamics taking into account these environmental and anthropogenic determinants (livestock management); (3) the modelling of the risk of pathogen transmission between wild and domestic animal populations. spatial for animal mobility simulation. The main methodological challenges that will be addressed are assimilation into multi-sensor Earth Observation data models, and mobility modelling at different scales.
Location : Zimbabwe, Mozambique
Partners : UMR TETIS/CIRAD, UMR ASTRE/CIRAD, UMR MIVEGEC/IRD, Faculty of Veterinary Science/University of Zimbabwe, Faculdade de veterinaria/Universidade Eduardo Mondlane
Financing : i-site MUSE, GEOSUD (image support)
Website: https://tempo.cirad.fr
ARBOCARTO Project: Dynamic spatial modelling of Aedes albopictus populations – Metropolitan France and Overseas Regions (2018)
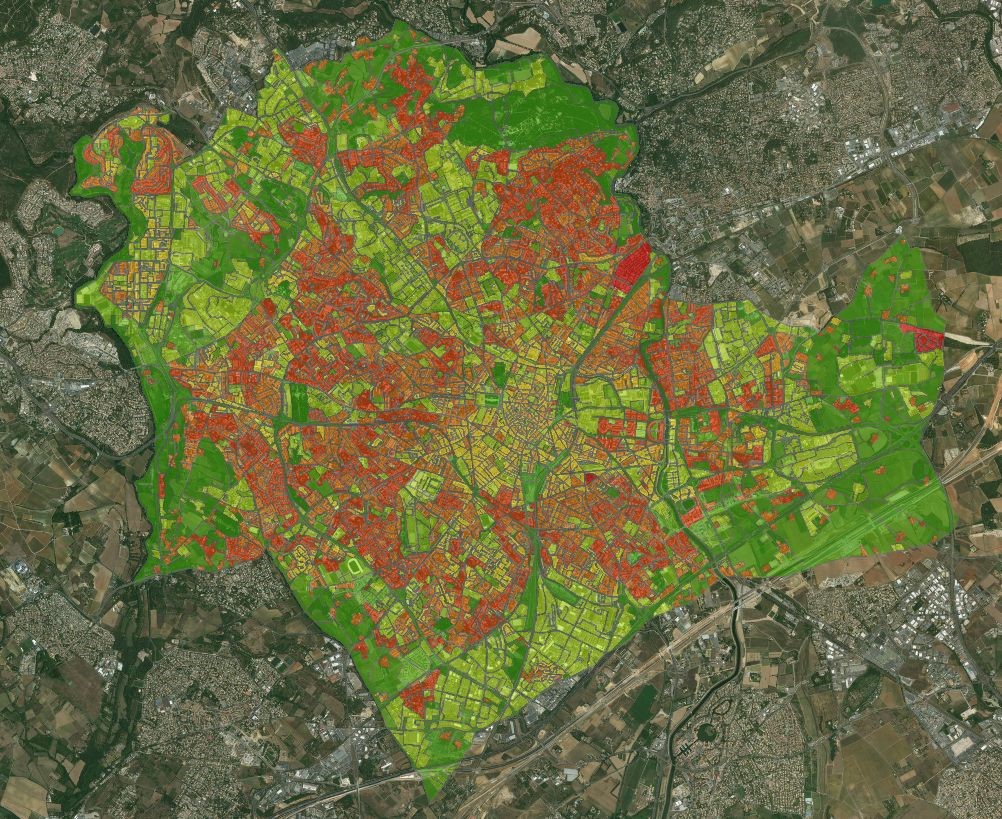
The Aedes albopictus tiger mosquito is one of the vectors of dengue, chikungunya and Zika viruses, and is fully adapted to urban environments. Aedes albopictus is present in the Indian Ocean (Reunion Island and Mayotte) and in many departments of metropolitan France, as well as in the West Indies and Guyana. The concomitant presence of vector mosquito and arbovirus populations is the cause of intense epidemics in the various overseas territories and constitutes an important risk also for France, where the tiger mosquito also constitutes a strong nuisance in the regions where it has recently established itself.
The objective of the Arbocarto project is to develop a predictive mapping tool for population densities of Aedes albopictus at a spatial scale adapted to the organization of surveillance and control actions carried out by vector control actors in different French departments, based on meteorological data (temperatures and daily rainfall) and remote sensing at high and very high spatial resolution.
Location : Metropolitan France and Overseas Regions
Partners : UMR TETIS/CIRAD, UMR Espace-Dev/IRD, Marie Demarchi, EID-Méditerranée, EID-Rhône Alpes, EID-Atlantique, ARS-Occitanie, CNES
Financing : Direction e de la Santé et Agences de Santé Auvergne Rhône Alpes et Nouvelle Aquitaine, GEOSUD (image support)
References
- Roux E., Tran A., Herbreteau V., Révillion C., Demarchi M., Mangeas M., Degenne P., Dehecq J.S. 2017. Space-time modelling of mosquito densities : application to Aedes albopictus, vector of viruses, in Reunion Island. GEOMED 2017, International Conference on Spatial Statistics Spatial Epidemiology and Spatial aspects of Public Health. Porto, Portugal, 7-9 Septembre
- Tran A., L’Ambert G., Lacour G., Benoît R., Demarchi M., Cros M., Cailly P., Aubry-Kientz M., Balenghien T., Ezanno P. 2013. A Rainfall- and Temperature-Driven Abundance Model for Aedes albopictus Populations. International Journal of Environmental Research and Public Health, 10 (5) : p. 1698-1719.
- Tran A., Herbreteau V., Demarchi M., Mangeas M., Roux E., Degenne P., Dehecq J.S. 2017. Spatial modeling of mosquito population dynamics : an operational tool for the surveillance of vector-borne diseases. 37th International Symposium on Remote Sensing of Environment (ISRSE-37) : Earth Observation for Development and Adaptation to a Changing World, 8-12 Mai, Tshwane (Afrique du Sud).
Evaluation of the contribution of multi-date and multi-sensor remote sensing in the construction of a spatialized indicator of malaria transmission risk in the Amazon region – TéléPal (05/2015-05/2017)
The project is structured in four tasks:
- Characterize, from optical data (Landsat 8 to 30 m, Spot 5 to 10 m, Pleiades 2 m) and radar (Sentinel 1 to 10 m, TerraSarX 1 m), the different environments favourable to malaria transmission.
- Develop new approaches to landscape analysis in order to highlight, through the construction of spatialized risk indicators and the use of entomological and epidemiological data, the interactions and risks of human-vector transmission in villages in the cross-border area.
- Evaluate the contribution of different satellite data sources (including altimetry satellites) to the characterization of vector habitat.
- Value the results.
Location: cross-border area French Guiana – Brazilian state of Amapa
Partners : UMR ESPACE-DEV/IRD, Amapá Central Laboratory of Epidemiology (LACEN-AP, Brazil), Amapá State National Research Institute (IEPA, Brazil), Parasitology Laboratory/Pasteur Institute of Guyana, Amazonian Ecosystems and Tropical Pathologies Associate Team (EpaT/UG), Laboratory of Geography, Environment and Health of the University of Brasilia (LAGAS/UnB), Climate and Health Observatory of the Oswaldo Cruz Foundation (Fiocruz), UMR LETG 6554 CNRS – COSTEL/CNRS.
Financing: Centre National d’Etudes Spatiales (CNES). GEOSUD (Image Support)
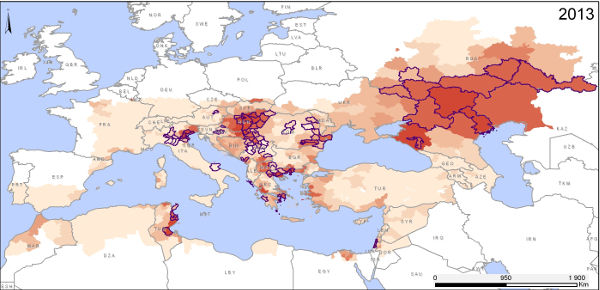
Use of MODIS imagery to map the risk of West Nile fever in Europe
The West Nile virus (WNV) is a pathogen transmitted by the bite of an infected mosquito. It is responsible for West Nile fever (WNF). The geographical distribution of this disease has changed dramatically over the last few decades, with the appearance of WNV for the first time in the Americas in 1999, and a recrudescence of cases since 2010 in numerous southern and eastern European countries, whereas the virus was previously present but not widely prevalent in Europe.
The WNV transmission cycle involves a bloodsucking insect (the vector), an animal host (the reservoir) and accidental hosts (horses and humans). Several different species of mosquito and wild birds can act as vectors and hosts.
The goal of a recent study published in the International Journal of Health Geographics was to examine the relationship between environmental variables and the occurrence of WNF outbreaks so as to map areas at risk from this disease within Europe.
The results of this study reveal the following risk factors:
- positive anomalies in a water index calculated from MODIS imagery for June,
- positive anomalies in temperature indices for July,
- the presence of wetlands,
- the presence of bird migration corridors,
- the occurrence of WNV outbreaks the previous year.
The predictive models constructed on the basis of epidemiological data reported between 2002 and 2011 were validated using data on WNF epidemics observed in 2012 and 2013, proving the model’s capacity to predict areas at risk of an outbreak of WNF from one year to the next. Generally speaking, this study has helped clarify the environmental and climatic determinants involved in the transmission of the WN virus in Europe, and highlights the role of satellite remote sensing in the setting up of public health surveillance systems including environmental monitoring.
For more information
Tran A., Sudre B., Paz S., Massimiliano R., Desbrosse A., Chevalier V., Semenza J., 2014. Environmental predictors of West Nile fever risk in Europe. International Journal of Health Geographics 13: 26. URL : http://www.ij-healthgeographics.com/content/13/1/26

Annelise Tran
Cirad | Tetis
@A.Tran
Contributions
Using SPOT imagery to plan ground truth surveys: application to the bio-ecological study of malaria vectors in French Guiana
Malaria is endemic in the French departments of Mayotte and Guiana. In the latter, an average of 3,450 people out of 230,000 to 300,000 inhabitants were affected by the disease in the 2000s. This number has decreased significantly since 2010, dropping below 1,000 cases for 2012, but transmission remains one of the highest in the Americas and its determinants remain largely unknown.
The Anopheles mosquito, certains species of which are responsible for transmitting the Plasmodium parasite at the origin of the disease to humans, must be regularly surveyed and their habitat characterised.
This need led to a study (1) on the bio-ecology of Anopheles mosquitoes in French Guiana. The village of Cacao was chosen for capturing adult mosquitoes and characterising their habitat. Previous records had shown the presence of a wide range of Anopheles species, it is an easily accessible area and contains a wide range of landscapes. The mosquito capturing technique (using human bait) is costly in terms of time and labour, and is restricted to a small number of capture sites. The study in question chose 12 sites in view of available resources.
In this context, a study published in BMC Ecology was designed to define and apply a method for choosing capture sites as objectively as possible, ensuring that they match environmental contexts which are significantly different from each other on the one hand, yet on the other, are typical of the region.
The method proposed first involved a detailed environmental characterisation of the study area, focusing on the river network, human impact, topography and land use. The data sources for this phase were varied, including maps and aerial photographs provided by IGN, the French mapping and survey agency, NASA’s digital elevation model (Shuttle Radar Topography Mission, SRTM) and SPOT 5 satellite images provided by the SEAS-Guiana direct receiving station.
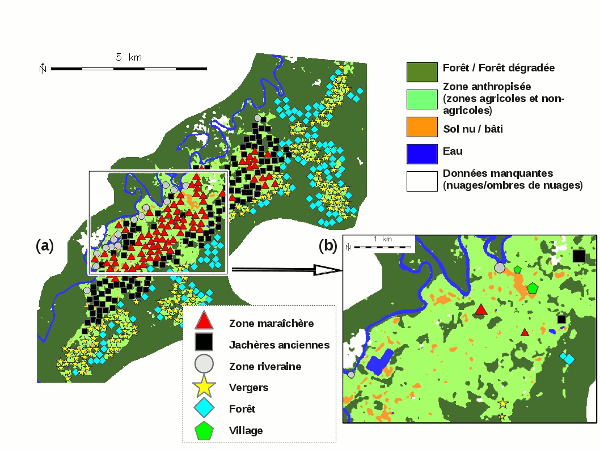
The second step involved identifying and characterising six distinct yet typical landscapes: the village, banks of the river Comté, market gardening areas in the plain, former fallow land, orchards on the slopes and hilltops and finally, dense forest. Two capture sites were chosen for each landscape profile taking into account accessibility.
The results of captures confirm the presence of a wide variety of Anopheles species (ten different species captured in all), including the main vector of malaria in Amazonia, Anopheles darlingi, and five secondary vector species. They also chose that, even in a relative small area of around 15 km x 5 km like at Cacao, the Anopheles species may be geographically structure and that land use—characterised by remote sensing techniques—is of great utility in explaining their distribution.
Further informations
Roux E., Gaborit P., Romaña C. A., Girod R., Dessay N., Dusfour I., 2013. Objective sampling design in a highly heterogeneous landscape – characterizing environmental determinants of malaria vector distribution in French Guiana, in the Amazonian region. BMC Ecology 2013, 13:45. URL: http://link.springer.com/article/10.1186/1472-6785-13-45
UMR228 Espace pour le Développement (Espace-Dev): http://www.espace-dev.fr/
Institut Pasteur de la Guyane : http://www.pasteur-cayenne.fr

Emmanuel Roux
IRD | UMR Espace-Dev
@E.Roux

Isabelle Dusfour
Institut Pasteur
idusfour@pasteur-cayenne.fr
(1) Study carried out under the research programme “Bioecology of Anopheles malaria vectors in Cacao, French Guiana: evaluating the risk of exposure to malaria and improving vector control”, subsidised by the State Secretariat for Overseas France and implemented by French Guiana’s Institut Pasteur in partnership with the Espace-Dev mixed research unit “Space for Development” (IRD, UAG, UM2, UR).
Thesis
- Extraction de relations spatio-temporelles à partir des données environnementales et de la santé
Hugo Alatrista-Salas
2013
Université Montpellier 2 - Modélisation du risque d’exposition aux moustiques vecteurs de Plasmodium spp. dans un contexte de lutte anti-vectorielle
Nicolas Moiraoux
2012
Université Montpellier 2 - Caractérisation des zones et périodes à risque de la Fièvre de la Vallée du Rift au Sénégal par télédétection et modélisation éco-épidémiologique
Valérie Soti
2011
Université Montpellier 2 - Utilisation de données d’observation de la terre par satellite pour l’évaluation des densités vectorielles et de la transmission du paludisme
Vanessa Machault
2010
Université Aix Marseille 2 - Le risque trypanosomien dans le bassin du Mouhoun au Burkina Faso : approches paysagères
Laure Guerrini
2009
Université Montpellier 3
Publications
Adde A., Roux E., Mangeas M., Dessay N., Nacher M., Dusfour I, Girod R. Briolant S. 2016. Dynamical Mapping of Anopheles darlingi Densities in a Residual Malaria Transmission Area of French Guiana by Using Remote Sensing and Meteorological Data. PLoS ONE, Public Library of Science, 11 (10)
Adde A., Dusfour I., Roux E., Girod R., Briolant S. 2016. Anopheles fauna of coastal Cayenne, French Guiana: modelling and mapping of species presence using remotely sensed land cover data. Mem Inst Oswaldo Cruz, 2016, 111 (12) pp.1 – 7
Amaral T.B., Gond V., Tran A.. 2016. Mapping the likelihood of foot-and-mouth disease introduction along the border between Brazil and Paraguay = Mapeamento do risco de introdução da febre aftosa na fronteira do Brasil com o Paraguai. Pesquisa Agropecuária Brasileira, 51 (5) : p. 661-670.
Blasdell K.R., Morand S., Henttonen H., Tran A., Buchy P.. 2016. Hantavirus seropositivity in rodents in relation to habitat heterogeneity in human-shaped landscapes of Southeast Asia. Spatial and Spatio-Temporal Epidemiology, 17 : p. 27-35.
Catry T., Li Z., Roux E., Herbreteau V., Révillion C., & Dessay N. 2016 | Fusion of SAR and Optical Imagery for Studying the Eco-Epidemiology of Vector-Borne Diseases in Tropical Countries. In Living Planet Symposium (Vol. 740, p. 8).
Catry T., Pottier A., Marti R., Li Z., Roux E., Herbreteau V., Mangeas M., Demagistri L., Gurgel H. et Dessay N. 2018 | Apports de la combinaison d’images satellites optique et RADAR dans l’étude des maladies à transmission vectorielle : cas du paludisme à la frontière Guyane française – Brésil, in Confins. https://journals.openedition.org/confins/15027
Della Rossa P., Tantrakarnapa K., Sutdan D., Kasetsinsombat K., Cosson J.F., Supputamongkol Y., Chaisiri K., Tran A., Supputamongkol S., Binot A., Lajaunie C., Morand S.. 2016 | Environmental factors and public health policy associated with human and rodent infection by leptospirosis: a land cover-based study in Nan province, Thailand. Epidemiology and Infection, 144 (7) : p. 1550-1562.
Girond F, Randrianasolo L, Randriamampionona L, Rakotomanana F, Randrianarivelojosia M, Ratsitorahina M, Brou T.Y., Herbreteau V., Mangeas M., Zigiumugabe S., Hedje J., Rogier C., Piola P., 2017 | Analysing trends and forecasting malaria epidemics in madagascar using a sentinel surveillance network: A web-based application. Malaria Journal 16:72.
Hammami P., Tran A., Kemp A., Tshikae P., Kgori P.M., Chevalier V., Paweska J., Jori F.. 2016 | Rift Valley fever vector diversity and impact of meteorological and environmental factors on Culex pipiens dynamics in the Okavango Delta, Botswana. Parasites and Vectors, 9 (434) : 17 p.
Herbreteau V., Révillion C., Trimaille E.. GeoHealth and QuickOSM, Two QGIS Plugins for Health Applications. In : Baghdadi N. (ed.), Mallet C. (ed.) Zribi M. QGIS and Generic Tools Volume 1 – QGIS in Remote Sensing. London: ISTE, 2018, (1), p. 257-286. (Earth Systems – Environmental Sciences: QGIS in Remote Sensing SET; 1). ISBN 978-1-78630-187-1
Herbreteau V. , Kassié D. , Roux E., Marti R., Catry T., Attoumane A., Révillion C., Commins J., Dessay N., Mangeas M. et Tran A. |Observer la Terre pour appréhender spatialement les inégalités de santé : regard historique et prospectif sur l’utilisation de la télédétection dans le domaine de la santé, in Confins 2018. https://journals.openedition.org/confins/15362
Ihantamalala F.A., Rakotoarimanana F.M.J., Ramiadantsoa T., Rakotondramanga J.M., Pennober G., Rakotomanana F., Cauchemez S., Metcalf C.J.E., Herbreteau V., Wesolowski A., 2018 | Spatial and temporal dynamics of malaria in Madagascar. Malaria Journal, 17, 58.
Li Z., Catry T., Dessay N., Roux E., Mahé E., & Seyler F. 2016 | Multi-sensor data fusion for identifying malaria environmental features. In IEEE International Geoscience and Remote Sensing Symposium (IGARSS), 2016 (pp. 2529-2532)
Li Z., Catry T., Dessay N., Roux E., & Seyler F. 2016 | Mapping soil typologies using geomorphologic features extracted from dem and SAR data: A environmental factor affecting malaria transmission in the Amazon. In IEEE International Geoscience and Remote Sensing Symposium (IGARSS), 2016 (pp. 3140-3143).
Li Z., Roux E., Dessay N., Girod R., Stefani A., Nacher M, Moiret A., Seyler F. 2016. Mapping a Knowledge-Based Malaria Hazard Index Related to Landscape Using Remote Sensing: Application to the Cross-Border Area between French Guiana and Brazil. Remote Sensing, MDPI, 8 (4): 22 p.
Moua Y., Roux E., Girod R., Dusfour I., De Thoisy B., Seyler F., Briolant S. 2016. Distribution of the Habitat Suitability of the Main Malaria Vector in French Guiana Using Maximum Entropy Modeling. Journal of Medical Entomology, Entomological Society of America, dec. 2016: 16 p.
Olive M.M., Chevalier V., Grosbois V., Tran A., Andriamandimby S.F., Durand B., Ravalohery J.P., Andriamamonjy S., Rakotomanana F., Rogier C., Héraud J.M.. 2016. Integrated analysis of environment, cattle and human serological data: Risks and mechanisms of transmission of rift valley fever in Madagascar. PLoS Neglected Tropical Diseases, 10 (7) : e0004827 (17 p.).
Randrianaivo H., Bertaut-Nativel B., André M,. Irabe M., Robillard PY., Boumahni B., Mangeas M., Roux E., Yao Brou T., Gérardin P., Filleul L., Herbreteau V., 2017. Mise en place d’une surveillance spatialisée des malformations congénitales à La Réunion : choix méthodologiques. Bull Epidémiol Hebd. (2):38-44.
Semenza J.C., Tran A., Espinosa L., Sudre B., Domanovic D., Paz S.. 2016. Climate change projections of West Nile virus infections in Europe: Implications for blood safety practices. Environmental Health, 15 (28) (suppl. 1. Healthy-Polis: Challenges and Opportunities for Urban Environmental Health and Sustainability) : 12 p..
http://dx.doi.org/10.1186/s12940-016-0105-4
Soti V., Tran A., Goebel F.R.. 2016. Potential of remote-sensing images to study the effect of natural vegetation on the spatial distribution of greyback canegrub, Dermolepida albohirtum, in Australia. In : Allsop Peter G. (ed.). 29th Congress of the International Society of Sugar Cane Technologists: abstracts book. Chiang Mai : Thailand Society of Sugar Cane Technologists, p. 120-121. ISSCT Congress. 29, 2016-12-05/2016-12-08, Chiang Mai (Thaïlande).
Tran A., Trevennec C., Lutwama J., Sserugga J., Gély M., Pittiglio C., Pinto J., Chevalier V.. 2016. Development and assessment of a geographic knowledge-based model for mapping suitable areas for Rift Valley fever transmission in Eastern Africa. PLoS Neglected Tropical Diseases, 10 (9) : e0004999 (20 p.).
Tran A., Kassié D., Herbreteau V. 2016. Applications of Remote Sensing to the Epidemiology of Infectious Diseases: Some Examples. In: Land Surface Remote Sensing: Environment and Risks, Baghdadi N. & Zribi M. (Eds). ISTE Press – Elsevier. 384 p.